
So the arrangement of molecules, in the long run, will remain the same. When an atom is missing or an atom is in an irregular place in the lattice structure, the corresponding defects are known as point defects. They have long term arrangement of their molecules.This means that they have repeated arrangement of a unit cell to form a space lattice structure. Meanwhile there exist many textbooks and monographs in this field of which I mention those of Read, Cottrell, Friedel. mes Sanglantes Defects Of A Crystal Lattice Country: Canada Released: Mar 2014 Genre: Electronic Style: Noise, Power Electronics, Experimental Notes. A line defect is restricted to a row of lattice points, and a plane defect involves an entire plane of lattice points in a crystal. This topic was treated by Seeger already in 19 in two Handbuch-articles of together about 500 pages. A point defect is any defect that involves only a single particle (a lattice point) or sometimes a very small set of points. 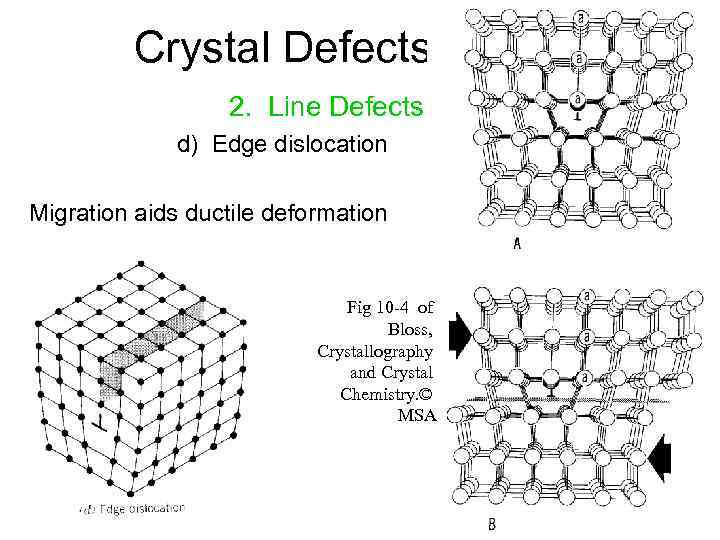
Frenkel defects are intrinsic defects because the existence causes the Gibbs energy of a crystal to decrease, which means it is favorable to occur.\): Edge Dislocations. 1 Introduction In the rst ve lectures, we have focussed our attentions on the methodologies used to describe crystal symmetry and on the scattering techniques employed to study the arrangement of atoms and spins in crystals (electronic, nuclear and spin densities). The most common types of Crystal defects are surface defects, substitutional defects, line defects, point defects, interstitial defects, Frenkel and Schottky. When we talk about crystalline solids, we say they have a definite structure. The present course of seven lectures is thought as an introduction into the theory of defects in crystal lattices. 
The introduction of defects in crystals is inevitable since they are already formed during the crystal growth phase. As interstitial element, hydrogen causes remarkable distortions in the crystal lattice, which predetermines the affinity of hydrogen atoms with vacancies, dislocations and grain boundaries. This often happens when a substance crystalizes too quickly. Where \(N\) is the number of normally occupied positions, \(N^*\) is the number of available positions for the moving ion, the \(\Delta H\) of formation is the enthalpy formation of one Frenkel defect, and \(R\) is the gas constant. Defects occur from both human error and the experimental setup. Crystal lattice defects are interruptions of the periodicity of the spatial arrangements of atoms. A lattice defect is a part of a crystal lattice where the repeating structure is disrupted.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
